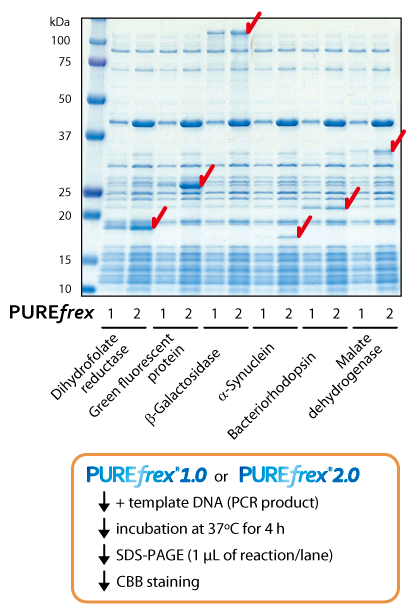
PUREfrex® ver. 1.0 vs 2.0
Overview
PUREfrex®1.0 was launched in 2011, now we improved the protein productivity of PUREfrex®1.0 and named the advanced version “PUREfrex®2.0”.
We modified the preparation methods of all components that were purified from E.coli and optimized the factors’ composition. As a result of this modification, PUREfrex®2.0 achieved 2-10 times higher protein productivity than PUREfrex®1.0.
Furthermore, the yield of active form of protein which needs a proper folding to active was increased when synthesized in PUREfrex®2.0 with chaperon and/or disulfide bond isomerase.
| PUREfrex®1.0 | PUREfrex®2.0 | |
|---|---|---|
| The amount of DHFR production per one kit (250 μL) | 40 μg | 150 μg |
| How many kits need for 100μg of DHFR production? | 660 μL (> 3kits) | 160 μL (< 1kit) |
| The amount of GFP production per one kit (250 μL) | 25 μg | 200 μg |
| How many kits need for 100μg of GFP production? | 1000 μL (> 4 kits) | 125 μL (< 1kit) |
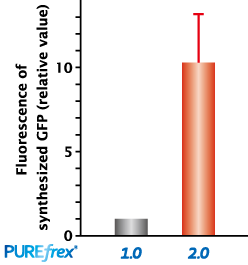
Compare the activity of synthesized protein
GFP
Fluorescence of synthesized with PUREfrex®2.0 is more than 10 times higher than PUREfrex®1.0.
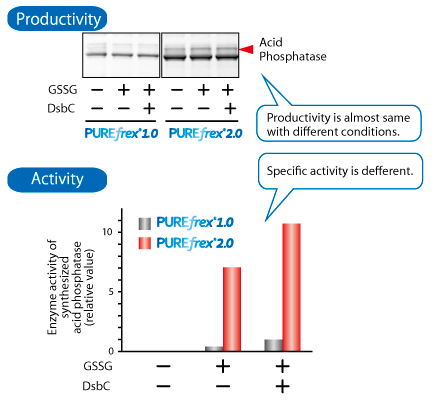
Acid phosphatase (AppA)
AppA from E.coli with 5 disulfide bonds, 1 of which is non-consecutive. The enzyme activity of synthesized AppA is increased when AppA synthesized with PUREfrex®2.0 and DsbC Set which contains disulfide bond isomerase DsbC.
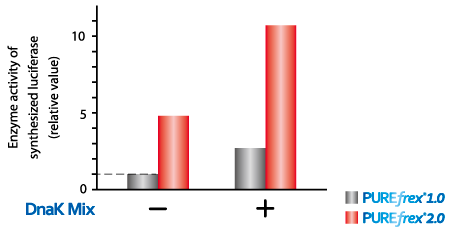
Luciferase
The enzyme activity if luciferase is increased when luciferase synthesized with PUREfrex®2.0 and Dnak mix which contains DnaK, DnaJ and GrpE.
“PUREfrex® is Registered in U.S. Patent and Trademark Office”